Activation reactions of 2-pyridyl and 2-pyrimidinyl alkynes with Ru3(CO)12†
Abstract
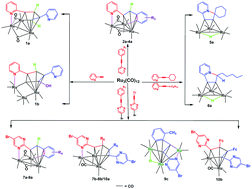
The reaction of Ru3(CO)12 with alkyne derivatives R1C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CR2 (R1 = 2-pyridyl, R2 = H (1), 2-CH3-Ph (2), 4-CH3O-Ph (3), 4-NO2-Ph (4), cyclohexyl (5), and n-hexyl (6); R1 = 5-Br-2-pyrimidinyl, R2 = Ph (7), 4-CH3O-Ph (8), 2-CH3-Ph (9), and ferrocenyl (10)) proceeds in toluene with the formation of triangular triruthenium clusters 1a–4a, 7a–9a, 7b–8b and 10a, linear triruthenium clusters 1b and 9c, Ru3(CO)9(μ-H)(μ3-η1:η2:η1-triruthenium) derivatives 5a–6a and ruthenole 10b. All compounds have been fully characterized by FT-IR and 1H NMR spectroscopy and ESI-MS, and most of them additionally by single-crystal X-ray diffraction. Each of the structures of 1a–4a, 7a–9a, 7b–8b and 10a has a triruthenium triangular skeleton containing a metallacyclopentadienyl framework formed by one ligand or two coupled ligands coordinating to two ruthenium atoms of the Ru3 moiety via a μ2-η2:η4 or μ4-η1:η2:η1:η1 bonding mode, and the linking of a pyridyl or a 5-Br-2-pyrimidinyl N atom with the third ruthenium atom. Each of the structures of 1b and 9c consists of an open, bent arrangement of three ruthenium atoms, with the organic moiety of 1b arising from the coupling of two ligands and of 9c being formed by one ligand through a Csp2–H activation in its benzene ring. Each of 5a–6a is formed by Csp3–H activation and bridging of the leaving hydrogen ion by a triruthenium carbonyl unit during the reactions of 5–6 and Ru3(CO)12. 10b is a common ruthenole. A detailed investigation on the reaction process showed that the strong coordination ability of the nitrogen atoms of 2-pyridyl and 5-Br-2-pyrimidinyl groups is more favorable for the formation of the products than the electronic and steric effects of the substituents in 1–10.
CR2 (R1 = 2-pyridyl, R2 = H (1), 2-CH3-Ph (2), 4-CH3O-Ph (3), 4-NO2-Ph (4), cyclohexyl (5), and n-hexyl (6); R1 = 5-Br-2-pyrimidinyl, R2 = Ph (7), 4-CH3O-Ph (8), 2-CH3-Ph (9), and ferrocenyl (10)) proceeds in toluene with the formation of triangular triruthenium clusters 1a–4a, 7a–9a, 7b–8b and 10a, linear triruthenium clusters 1b and 9c, Ru3(CO)9(μ-H)(μ3-η1:η2:η1-triruthenium) derivatives 5a–6a and ruthenole 10b. All compounds have been fully characterized by FT-IR and 1H NMR spectroscopy and ESI-MS, and most of them additionally by single-crystal X-ray diffraction. Each of the structures of 1a–4a, 7a–9a, 7b–8b and 10a has a triruthenium triangular skeleton containing a metallacyclopentadienyl framework formed by one ligand or two coupled ligands coordinating to two ruthenium atoms of the Ru3 moiety via a μ2-η2:η4 or μ4-η1:η2:η1:η1 bonding mode, and the linking of a pyridyl or a 5-Br-2-pyrimidinyl N atom with the third ruthenium atom. Each of the structures of 1b and 9c consists of an open, bent arrangement of three ruthenium atoms, with the organic moiety of 1b arising from the coupling of two ligands and of 9c being formed by one ligand through a Csp2–H activation in its benzene ring. Each of 5a–6a is formed by Csp3–H activation and bridging of the leaving hydrogen ion by a triruthenium carbonyl unit during the reactions of 5–6 and Ru3(CO)12. 10b is a common ruthenole. A detailed investigation on the reaction process showed that the strong coordination ability of the nitrogen atoms of 2-pyridyl and 5-Br-2-pyrimidinyl groups is more favorable for the formation of the products than the electronic and steric effects of the substituents in 1–10.


 Please wait while we load your content...
Please wait while we load your content...