Three-dimensional porphyrin-based covalent organic frameworks with tetrahedral building blocks for single-site catalysis†
Abstract
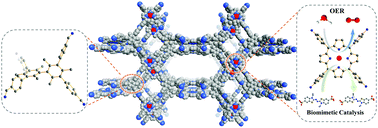
Two three-dimensional (3D) porphyrin covalent organic frameworks, PCOF-1 and PCOF-2, were synthesized via imine condensation of a planar porphyrin tetramine (TAPP or TABPP) and a rigid tetrahedral aldehyde based on the steric hindrance of 3,3′,5,5′-tetrakis(4-formylphenyl)bimesityl (TFBM). The structures of PCOF-1 and PCOF-2 were studied by FT-IR and 13C CP-MAS solid state NMR spectroscopy. Powder X-ray diffraction patterns revealed obvious crystallinity with two intense peaks at 3.28° and 3.75° for PCOF-1, and 2.63° and 2.98° for PCOF-2. Structural simulation confirmed their 3D rutile type (pts) topological structures with two different pore sizes. X-ray single crystal diffraction revealed a distorted tetrahedral structure for the building block TFBM with two dihedral angles of 119° and 107.8°, and a planar square structure for the model compound (MC) with an outspread angle of 176.5°. PCOF-1 and PCOF-2 exhibited Brunauer–Emmett–Teller (BET) surface areas of 316 and 234 m2 g−1, respectively. The morphologies of PCOF-1 and PCOF-2 were investigated by scanning electron microscopy and transmission electron microscopy methods. PCOF-1 and PCOF-2 showed a high thermal stability up to 420 °C without decomposition through thermogravimetric analysis (TGA), and high chemical stability with no obvious mass loss after three days of immersion in various solutions. Due to the large surface area and the appropriate pore size, PCOF-Fe exhibited excellent biocatalytic catalytic performance, while PCOF-Co exhibited good electrocatalytic activity towards oxygen evolution reactions. These results indicate that 3D porphyrin-based COFs constructed from the tetrahedral building block with steric hindrance are promising candidates for single-site catalysis.


 Please wait while we load your content...
Please wait while we load your content...