Visual detection of H2O2 and melamine based on PW11MO39n− (M = Cu2+, Co2+, Mn2+, Fe3+) and PW9M3O34n− (M = Cu2+, Co2+, Mn2+, Fe3+)†
Abstract
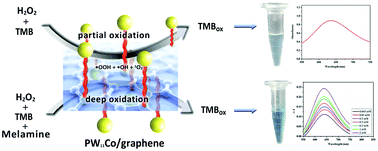
The peroxidase-like activity of transition metal-substituted polyoxometalates (POMs) (PW11MO39n−, PW9M3O34n−, abbreviated as PW11M and PW9M3, where M = Cu2+, Co2+, Mn2+, Fe3+) in the oxidation of 3,3′,5,5′-tetramethylbenzidine (TMB) by H2O2 was evaluated. Among all POMs, PW11Cu and PW9Cu3 were the most active due to their high redox potentials. Based on these findings, H2O2 could be detected through observation of TMB/UV-Vis signals in a wide pH range of 2.0–8.0 with linear ranges of 8.96 × 10−5–2.2 × 10−3 mM at a detection limit of 6 nM and 1.21 × 10−5–1.21 × 10−3 mM at a detection limit of 0.98 nM corresponding to PW11Cu and PW9Cu3, respectively. Raman and electron paramagnetic resonance measurements revealed that melamine addition could improve the peroxidase-like activity of PW11Co (Cu) because of the interaction between PW11Co (Cu) and melamine to generate several OH· radicals. Thus, a novel method for the colorimetric detection of melamine was developed at a detection limit as low as 0.1 and 1.5 μM based on PW11Co using UV-Vis spectroscopy and naked eye observation in a wide pH range of 2.0–8.0. The TMB/H2O2/PW11Co system was essentially used for melamine detection in liquid or powder milk, which showed a promising route to construct POM peroxidase-like mimics in the colorimetric detection of H2O2 and melamine with a cost-effective and simple synthesis mechanism instead of enzyme and noble metal utilization. PW11Co/graphene enhanced the peroxidase-like activity of PW11Co in the colorimetric detection of melamine to 0.02 μM detection limit with a linear range of 2 × 10−4–8 × 10−3 mM and it can be recycled due to its heterogeneity.


 Please wait while we load your content...
Please wait while we load your content...