A maltoheptaose-decorated BODIPY photosensitizer for photodynamic inactivation of Gram-positive bacteria†
Abstract
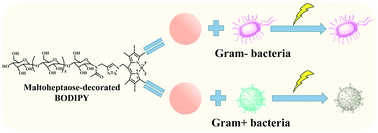
Due to the occurrence of bacterial resistance, there is a significant need to develop alternative antimicrobials to conventional antibiotics. Antibacterial photodynamic therapy (PDT) has been recognized as a promising antibacterial strategy with a low likelihood of generating bacterial resistance. In this work, a maltoheptaose-decorated boron-dipyrromethene (BODIPY) photosensitizer (BODIPY-Mal-I) was synthesized via a copper(I)-catalyzed azide alkyne cycloaddition (CuAAC) reaction, followed by diiodo substitution at the 2,6-positions of the BODIPY core. The as-obtained BODIPY-Mal-I, consisting of a hydrophobic 2,6-diiodo-tetramethyl BODIPY unit and a hydrophilic maltoheptaose segment, can self-assemble and form microaggregates in aqueous medium. Under light illumination, BODIPY-Mal-I in water could generate singlet oxygen rapidly. The generated singlet oxygen could kill the planktonic cells of Gram-positive bacteria efficiently. BODIPY-Mal-I could also eradicate early-stage biofilms of Gram-positive bacteria in the presence of light illumination. In addition, BODIPY-Mal-I exhibits low cytotoxicity to zebrafish embryo without light illumination.


 Please wait while we load your content...
Please wait while we load your content...