Probing the influence of lactose fines, a USP modified induction port and modified DDU apparatus on the aerodynamic behavior of a fluticasone propionate dry powder inhaler†
Abstract
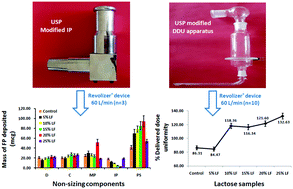
The objective of the present study is to understand the effects of lactose fines, a USP modified induction port (IP) and USP modified glass sampling apparatus (DDU apparatus) on the aerodynamic behavior of a fluticasone propionate (FP) dry powder inhaler (DPI). Lactose carriers containing 0 to 25% lactose fines were assessed for various physicochemical properties. Also, formulated FP DPIs were analyzed in terms of aerodynamic behavior using a multistage cascade assembly and DDU apparatus equipped with Revolizer® at a flow rate of 60 L min−1. The physicochemical characterization of lactose carriers showed that there were remarkable differences in particle size distribution, surface area and surface roughness. The formulated FP DPIs exhibited acceptable percentage content uniformity (>96%). A cascade impactor equipped with the USP modified IP showed significant differences in recovered and emitted doses compared to the USP IP. Additionally, in comparison to the USP IP, the USP modified IP showed a lower amount of drug deposition within the IP, irrespective of the lactose fines concentration. Besides, a formulated FP DPI showed a higher (>95%) mean delivered dose using the modified DDU apparatus at a flow rate of 60 L min−1. Briefly, not only the formulation properties and inhalation device but also the IP geometry had significant impacts on the in vitro analysis. The present investigation is the first detailed account of FP aerosolization performance using USP modified IP and DDU apparatus. Therefore, these findings may be of high importance for both academics and industrial researchers.


 Please wait while we load your content...
Please wait while we load your content...