Adsorption of charged anisotropic nanoparticles at oil–water interfaces†
Abstract
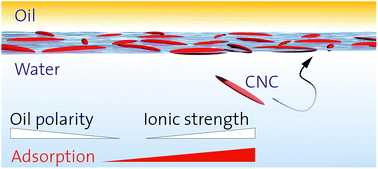
The adsorption of nanoparticles at fluid interfaces is of profound importance in the field of nanotechnology. Recent developments aim at pushing the boundaries beyond spherical model particles towards more complex shapes and surface chemistries, with particular interest in particles of biological origin. Here, we report on the adsorption of charged, shape-anisotropic cellulose nanocrystals (CNCs) for a wide range of oils with varying chemical structure and polarity. CNC adsorption was found to be independent of the chain length of aliphatic n-alkanes, but strongly dependent on oil polarity. Surface pressures decreased for more polar oils due to lower particle adsorption energies. Nanoparticles were increasingly wetted by polar oils, and interparticle Coulomb interactions across the oil phase thus increase in importance. No surface pressure was measurable and the O/W emulsification capacity ceased for the most polar octanol, suggesting limited CNC adsorption. Further, salt-induced charge screening enhanced CNC adsorption and surface coverage due to lower interparticle and particle–interface electrostatic repulsion. An empiric power law is presented which predicts the induced surface pressure of charged nanoparticles based on the specific oil–water interface tension.


 Please wait while we load your content...
Please wait while we load your content...