Selective electrochemical CO2 conversion to multicarbon alcohols on highly efficient N-doped porous carbon-supported Cu catalysts†
Abstract
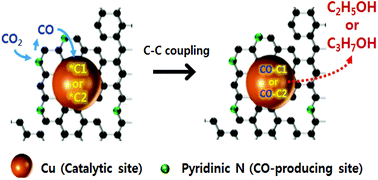
The selective and efficient electrocatalytic transformation of carbon dioxide (CO2) to multicarbon alcohols (e.g., C2H5OH and C3H7OH) is a challenge in renewable and sustainable energy research. Herein, a series of hybrid catalysts consisting of Cu nanoparticles supported on N-doped porous carbon (Cu/NPC) were prepared. It was demonstrated that the selectivity for C2H5OH or C3H7OH could be tuned by introducing N-doped porous carbon materials as cocatalysts with different pyridinic N contents, which could in situ produce a reactive CO intermediate from CO2. By varying the pyridinic N content, highly selective production of multicarbon alcohols was achieved using the Cu/NPC hybrid catalysts with a high faradaic efficiency for one pot production of multicarbon alcohols up to 73.3% at −1.05 V (vs. RHE). The faradaic efficiency for C2H5OH and C3H7OH was 64.6% and 8.7%, respectively. The pyridinic N species were likely the CO-producing sites and together with Cu catalytic sites acted cooperatively to produce C2H5OH and C3H7OH via a two-site mechanism for efficient CO2 reduction to multicarbon alcohols. These findings provide novel guidance for the rational design of electrocatalysts and for tuning the catalytic activity and selectivity for multicarbon alcohol production from CO2.


 Please wait while we load your content...
Please wait while we load your content...