Supercritical methanol depolymerization and hydrodeoxygenation of lignin and biomass over reduced copper porous metal oxides†
Abstract
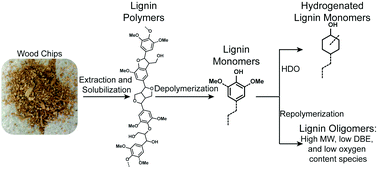
Supercritical methanol depolymerization and hydrodeoxygenation (SCM-DHDO) of maple wood and lignin extracted with GVL from maple wood, was studied using a copper porous metal oxide catalyst. Phenols (P or H), guaiacols (G), and syringols (S) with deoxygenated 1 to 3-carbon (C1–C3) alkyl tails were the early products (15 min reaction time) from both the maple wood and the GVL extracted lignin. Furthermore, at 15 min reaction time, the maple wood products showed increased levels of demethoxylation with an S/G/P ratio of 1.0/4.2/1.2 versus 1.0/1.0/0.1 from the GVL extracted lignin products. After 4 h of reacting lignin monomers, dimers and trimers were produced with both the maple wood and extracted lignin. However, the 4 h maple wood products yielded twice the amount of lignin monomers (43.1%) as compared to the 4 h GVL extracted lignin products (20.1%). The GVL extracted lignin products were in the dimer to trimer molecular weight (MW, ∼180–750 Da) ranges whereas the maple wood products were in the monomer to dimer MW ranges (∼94–500 Da) according to the GPC results. NMR and FT-ICR MS analyses show that both the 4 h maple wood products and 4 h GVL extracted lignin products undergo a high degree of hydrodeoxygenation, saturation, and repolymerization by C–C bond formation. The higher yield of lignin monomers from the maple wood is likely due to the lower lignin concentration that occurs during the reaction whereas higher lignin concentration during the reaction leads to more oligomerization reactions.


 Please wait while we load your content...
Please wait while we load your content...