endo- versus exo-Cyclic coordination in copper complexes with methylthiazolylcarboxylate tacn derivatives†
Abstract
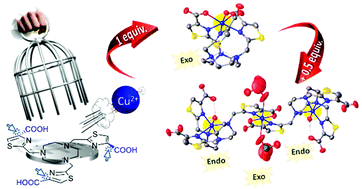
Three tacn (1,4,7-triazacyclononane)-based ligands substituted by methylthiazolylcarboxylate (tha) and/or methylthiazolyl (th) arms have been examined for copper complexation with the aim to study the impact of carboxylate groups on the complexation of Cu(II), which can present an endo- or exo-cyclic coordination. Two new ligands have been synthesised: H3no3tha, tacn bearing three methylthiazolylcarboxylate arms, and H2no1th2tha, tacn with one methylthiazolyl and two methylthiazolylcarboxylate arms, while Hno2th1tha had already been described. Their complexation behaviour with 1 or 1.5 equivalents of metal was studied on the basis of preliminary results showing the tendency of tha arms to form exocyclic polynuclear species. The solid state studies of the Cu(II) and Zn(II) complexes were investigated and some of their structures were characterised by X-ray diffraction. The physicochemical properties of the complexes in solution were also investigated by means of potentiometric measurements, UV-vis spectroscopy, EPR and computational studies, NMR characterisation of the corresponding Zn(II) complexes and redox behaviour by electrochemistry. Mono- and tri-nuclear complexes ML and M3L2 were formed and isolated, highlighting the tendency of methylthiazolylcarboxylate arms, when carried by a tacn platform, to form exo-cyclic and polynuclear complexes. However, this exhaustive study evidences that the “out of cage” and “in cage” present different behaviour in terms of stability.
- This article is part of the themed collection: Nitrogen Ligands


 Please wait while we load your content...
Please wait while we load your content...