Non-uniform electron conduction in weakly ordered SrFe1−xMoxO3−δ
Abstract
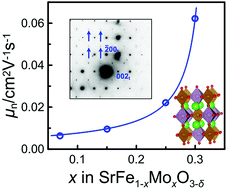
The electrical conductivity of SrFe1−xMoxO3−δ (0.07, 0.15, 0.25) was measured in the range of oxygen partial pressure of 10−16–0.5 atm and at temperatures 800–950 °C by a four-probe dc technique. Experimental results were satisfactorily simulated with a model suggesting that oxygen ions and electronic charge carriers of n- and p-types were involved in conduction. The mobility of charge carriers was calculated using partial conductivities and earlier published oxygen nonstoichiometry data. The mobility of p-type charge carriers was found to decrease in response to a decreasing oxygen content or an increasing molybdenum content in the oxide. The mobility of n-type carriers was found to be unaffected by the oxygen content, but exhibited an accelerating increase upon increasing the molybdenum content. Such behavior of the electron mobility was interpreted in view of the tendency of iron and molybdenum cations to undergo ordering based on the supposition that two different mechanisms of electron transport were involved in these oxides. It was assumed that nanoscale ordered areas with fast electron transport dispersed in the disordered perovskite matrix played the role of a high-conductivity filler in a composite consisting of two components with different conductivities. The behavior of the effective electron mobility was approximated well using the percolation theory. The molybdenum content x = 0.327 was calculated to be the percolation threshold in SrFe1−xMoxO3−δ.


 Please wait while we load your content...
Please wait while we load your content...