On the active site for electrocatalytic water splitting on late transition metals embedded in graphene†
Abstract
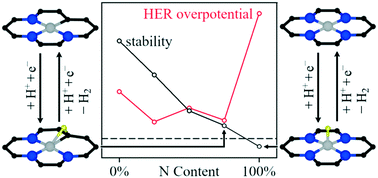
Transition-metal atoms embedded in nitrogen-doped graphene can be used for electrocatalytic water splitting, but there are open questions regarding the identity of the active site. We study the formation of hydrogen and oxygen as well as the reduction of oxygen on 14 transition metals embedded in nitrogen-doped graphene using density functional theory and find that the stability and the catalytic properties of the metals depend on the nitrogen content of the support. While previous studies focus on metal atoms inside nitrogen-free and fully substituted vacancies, we find that partially nitrogen-substituted single sites are significantly more active for later transition metals (groups 10–11). These sites are also more stable than previously suggested active-site models. Our findings suggest that stability and catalytic activity of late transition metals embedded in graphene could be increased by controlling the nitrogen content of the support to obtain partially substituted vacancy sites. For early transition metals, fully substituted vacancies represent the most active site.


 Please wait while we load your content...
Please wait while we load your content...
