Highly improved acetone oxidation activity over mesoporous hollow nanospherical MnxCo3−xO4 solid solutions†
Abstract
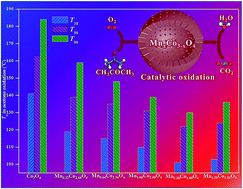
Hierarchical bimetal oxide solid solutions with mesoporous hollow-interior architecture have greatly promising applications in environmental catalysis due to their exceptional catalytic performances, while facile synthesis of such a unique architecture is still challenging. Herein, mesoporous hollow nanospherical (MHS) MnxCo3−xO4 solid solutions with tunable Mn/Co molar ratios in the spinel-type cubic crystal phase, fabricated by a facile solvothermal alcoholysis approach, have been developed to catalyze acetone oxidation for the first time. Experimental results confirm that MHS Mn1.20Co1.80O4 gave the optimal acetone oxidation activity, achieving 100% acetone conversion and 100% CO2 selectivity at 140 °C under the reaction conditions of acetone concentration = 1000 ppm, 20%O2/N2 and WHSV = ca. 93 000 mL gcat−1 h−1. This superior activity of MHS Mn1.20Co1.80O4 can be mainly ascribed to the strong synergetic effect of the unique mesoporous hollow nanospherical feature, abundant oxygen vacancies and surface active oxygen species, the enhanced chemical nature resulting from the presence of more Mn4+, Mn3+ and Co3+ reactive sites, and the improved redox capacities. Kinetics studies provide direct evidence to validate the finding that the strong synergetic effect rather than individual factors played the critical role in determining the acetone oxidation activity. Meanwhile, a plausible catalytic mechanism of acetone oxidation over MHS MnxCo3−xO4 solid solutions has been proposed and elaborated in detail by elucidating the structure–activity relationship. Furthermore, MHS Mn1.20Co1.80O4 also presented excellent long-term stability and good water tolerance due to its highly stable crystal phase and robust morphological structure, showing good potential in eliminating volatile organic compounds.


 Please wait while we load your content...
Please wait while we load your content...