Electricity-driven 7α-hydroxylation of a steroid catalyzed by a cytochrome P450 monooxygenase in engineered yeast†
Abstract
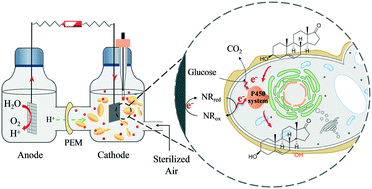
NAD(P)H are the cofactors of the cytochrome P450 monooxygenases for the catalytic hydroxylation of steroids, which is notoriously challenging for traditional synthetic chemistry due to the requirement of specificity and chirality. However, an undersupply of NAD(P)H is a crucial bottleneck in restricting the conversion rate of P450-catalyzed steroid hydroxylation. To address the problem of cofactor undersupply and enhance the biotransformation from dehydroepiandrosterone (DHEA) to 7α-OH-DHEA, an electricity-driven NAD(P)H regeneration shortcut was incorporated into recombinant Saccharomyces cerevisiae harbouring 7α-hydroxylase (CYP7B1) to construct a P450 bioelectrocatalytic system (BES). The BES could collaboratively utilize electrons derived from both electrodes and the oxidation of carbon sources, enabling intracellular NAD(P)H regeneration via the concomitant electron shuttle (i.e., neutral red (NR))-mediated extracellular electron transfer (EET) pathway and the oxidation of glucose to facilitate the P450 enzyme-catalysed hydroxylation of steroids. Thus, the 7α-OH-DHEA yield reached 288.6 ± 7.8 mg L−1 in the BES, ∼2.4-fold its counterpart in the absence of EET (122.1 ± 3.7 mg L−1). For the first time, a NR-mediated BES was exhibited to be an efficient strategy to enhance steroid hydroxylation in engineered yeast, being promising for steroid conversion in pharmaceutical engineering.
- This article is part of the themed collection: 2019 Catalysis Science & Technology HOT Articles


 Please wait while we load your content...
Please wait while we load your content...