One catalyst, multiple processes: ligand effects on chemoselective control in Ru-catalyzed anti-Markovnikov reductive hydration of terminal alkynes†
Abstract
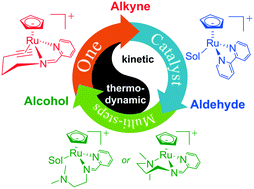
Cost-efficient ruthenium complexes are prominent catalysts for many important chemical processes, leading to a challenge in chemoselective control for Ru catalysis. A series of half-sandwich ruthenium(II) complexes with diverse N,N-bidentate ligands showed intriguing chemoselectivity in the catalyzed reductive hydration of alkynes. To reveal the ligand effects on chemoselective control, DFT calculations were performed to systematically study their detailed mechanisms. Results indicate that the pendant alkyne on catalyst A coordinated with ruthenium hinders the hydration reaction due to unfavored ligand exchange with substrates. In contrast, catalyst B without a pendant group can catalyze the hydration of alkynes. However, catalyst B fails to further hydrogenate the aldehyde product into an alcohol, due to the lack of a bifunctional type of catalysis. With a pendant amine substituent on N,N-bidentate ligands, catalysts C and D can catalyze both the hydration of the alkyne and the hydrogenation of the aldehyde into an alcohol. The terminal amine ligand is advantageous to both the ligand exchange step and the bifunctional hydrogenation step. Our studies unfold the unique outer-sphere vinylidene mechanism for the Ru-catalyzed reductive hydration of terminal alkynes, which well explains the observed anti-Markovnikov hydration in experiments. These mechanistic insights should provide useful guidelines for the rational design of more efficient chemoselective single-catalyst systems for multiple catalytic processes.


 Please wait while we load your content...
Please wait while we load your content...