Physical conditions for UO formation in laser-produced uranium plumes
Abstract
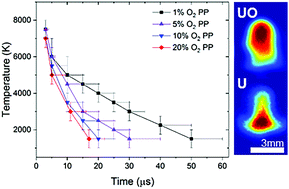
We investigate the oxidation of uranium (U) species, the physical conditions leading to uranium monoxide (UO) formation and the interplay between plume hydrodynamics and plasma chemistry in a laser-produced U plasma. Plasmas are produced by ablation of metallic U using nanosecond laser pulses. An ambient gas environment with varying oxygen partial pressures in 100 Torr inert Ar gas is used for controlling the plasma oxidation chemistry. Optical emission spectroscopic analysis of U atomic and monoxide species shows a reduction in the emission intensity and persistence with increasing oxygen partial pressure. Spectral modelling is used for identifying the physical conditions in the plasma that favor UO formation. The optimal temperature for UO formation is found to be in the temperature range of ∼1500–5000 K. The spectrally integrated and spectrally filtered (monochromatic) imaging of U atomic and molecular species reveals the evolutionary paths of various species in the plasma. Our results also highlight that oxidation in U plasmas predominantly occurs at the cooler periphery and is delayed with respect to plasma formation, and the dissipation of molecular species strongly depends on oxygen partial pressure.


 Please wait while we load your content...
Please wait while we load your content...