Hydrogen atom attachment to histidine and tryptophan containing peptides in the gas phase
Abstract
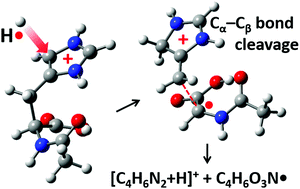
In this study, we use a combination of tandem mass spectrometry and hydrogen radical-mediated fragmentation techniques to analyze the sequence of peptides. We focus on fragmentation induced by the attachment of hydrogen atoms to the histidine and tryptophan residue side-chains in the peptide that occurs in the gas-phase. The hydrogen atom attached to the imidazole and indole rings in the histidine and tryptophan residues, respectively, and the resulting intermediate experienced Cα–Cβ bond cleavage. The detailed fragmentation mechanism is investigated by computational analysis using density functional theory. According to the results, hydrogen attachment occurs at the C-5 position in histidine and at the C-2 position in the tryptophan, which has a lower activation energy compared with the other positions and the resulting intermediate radicals yielded fragments due to Cα–Cβ bond cleavage. For the peptides that contain the histidine and tryptophan residues, cleavages in the Cα–Cβ and N–Cα bonds occurred independently. Therefore, the method presented in this study is applicable when analyzing peptides that contain histidine and tryptophan residues.
- This article is part of the themed collection: 2019 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...