Structure and reactivity of 2,4,6-tricyano-1,3,5-triazine under high-pressure conditions†
Abstract
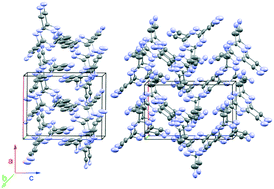
2,4,6-Tricyano-1,3,5-triazine is an attractive precursor for the synthesis of extended layered and nanoporous carbon nitrides. Using high pressure can achieve this via one-step condensation reactions. As a first step towards this goal we have characterized the structural properties and stability P–T range of the monomer (phase I) using synchrotron X-ray diffraction combined with FTIR spectroscopy under combined high pressure and high temperature conditions in a resistively heated diamond anvil cell. A new high pressure structure (phase II) appeared following compression to above 2.4 GPa at ambient temperature. The structure was identified as orthorhombic belonging to space group P212121. The pressure–temperature conditions of the I–II phase boundary, as well as the melting line have been established. The chemical transformation of the phase II crystal was studied between 4 and 10 GPa at temperatures ranging from 550 to 300 K. X-ray and spectroscopic examination of the reaction products revealed a crystalline phase that could be assigned to a layered carbon nitride containing linked s-triazine rings along with amorphous material. Analysis of the kinetics indicates that the structural transformation is driven by defect formation.
- This article is part of the themed collection: The effects of extreme conditions on molecular solids


 Please wait while we load your content...
Please wait while we load your content...