Determination of phenol degradation in chloride ion rich water by ferrate using a chromatographic method in combination with on-line mass spectrometry analysis†
Abstract
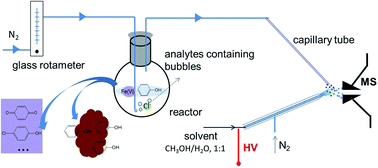
The degradation of phenol in Cl−-rich water by Fe(VI) was investigated using both chromatographic methods (HPLC and IC) and an on-line direct mass spectrometry (MS) analysis technique. Participation of Cl− during phenol degradation was observed through the formation of p-chlorophenol and coupled species with phenol. Though only a small concentration of Cl− was involved in the reaction, the presence of Cl− could affect the behavior of phenol substantially. With the increase of the Cl− concentration, generation of p-benzoquinone and p-chlorophenol was increased. Meanwhile, phenol in the bulk solution displayed a sharp decline followed by a gradual increase. The developed on-line MS analysis facilitated real-time monitoring of phenol changes in the reaction system. The results showed that phenol degradation by Fe(VI) with the presence of a high concentration of Cl− followed a second-order reaction process. The degradation pathway of phenol was also identified, with p-chlorophenol, p-benzoquinone, maleic acid and formic acid as the main byproducts.


 Please wait while we load your content...
Please wait while we load your content...