Phase transformations and capacity fade mechanism in LixSn nanoparticle electrodes revealed by operando 7Li NMR†
Abstract
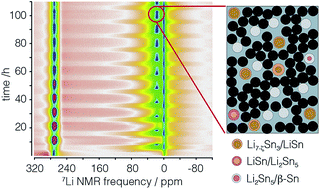
Operando 7Li nuclear magnetic resonance (NMR), ex situ7Li magic-angle spinning (MAS) NMR and pair distribution function (PDF) methods are used to investigate the electrochemical lithiation and delithiation of 60 nm particles of tin. While the structural transformation pathways between Li–Sn intermetallics during lithiation and delithiation of Sn nanoparticles are somewhat consistent with the structural evolution of Li–Sn phases expected from the equilibrium binary phase diagram, there are some notable exceptions with the observation of a metastable phase Li2Sn3, and two vacancy rich metastable phases, Li7−ζSn3, and Li13−δSn5 during delithiation. The capacity fade of ∼30% per cycle is not attributed to particle cracking, but rather to particles losing contact with the carbon/PVDF binder. The disconnecting particles are associated with LixSn phases undergoing large decreases in diameters on delithiation, i.e., Sn, Li2Sn5, LiSn, and Li7Sn3. A peculiar behavior of capacity fade in this system is that it only occurs during delithiation. This is explained by a model of disconnecting–reconnecting LixSn-containing particles which remain in the electrode and become reconnected every cycle by the volume increase of neighboring connected particles during the lithiation process.


 Please wait while we load your content...
Please wait while we load your content...
