Recent advances on the thermal destabilization of Mg-based hydrogen storage materials
Abstract
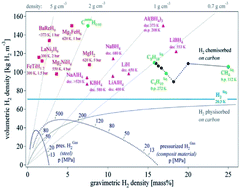
Magnesium hydride and its compounds have a high hydrogen storage capacity and are inexpensive, and thus have been considered as one of the most promising hydrogen storage materials for on-board applications. Nevertheless, Mg/MgH2 systems suffer from great drawbacks in terms of kinetics and thermodynamics for hydrogen uptake/release. Over the past decades, although significant progress has been achieved with respect to hydrogen sorption kinetics in Mg/MgH2 systems, their high thermal stability remains the main drawback, which hinders their practical applications. Accordingly, herein, we present a brief summary of the synthetic routes and a comprehensive overview of the advantages and disadvantages of the promising strategies to effectively tune the thermodynamics of Mg-based materials, such as alloying, nanostructuring, metastable phase formation, changing reaction pathway, and nano Mg-based composites. Among them nanostructuring and metastable phase formation, which have the superiority of changing the thermodynamics without affecting the hydrogen capacity, have attracted increasing interest in this field. To further optimize the hydrogen storage performance, we specially emphasize novel nanostructured materials, which have the advantage of combining alloy engineering, nanostructuring and the synergistic effect to change the thermodynamics of Mg/MgH2 to some extent. Furthermore, the remaining challenges and the directions of further research on MgH2, including the fundamental mechanism of the Mg–H bond instability, advanced synthetic routes, stabilizing nanostructures, and predicting novel composite materials, are proposed.


 Please wait while we load your content...
Please wait while we load your content...