Furan-flanked diketopyrrolopyrrole-based chalcogenophene copolymers with siloxane hybrid side chains for organic field-effect transistors†
Abstract
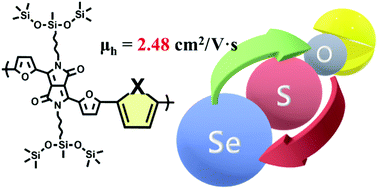
Herein, we report the effects of chalcogen atoms on organic field-effect transistors (OFETs) by studying a series of furan-flanked diketopyrrolopyrrole copolymers (PFDPPF-Si, PFDPPT-Si, and PFDPPS-Si) with different chalcogenophene comonomers (furan, thiophene, and selenophene), where the siloxane-terminated chains are used as solubilizing groups. The optical/electrochemical properties, microstructural analyses, and charge transport characteristics of the resulting copolymers are discussed as a function of the different chalcogenophenes. A larger heteroatom size led to a bathochromic shift in the absorption profiles and narrowing of the band gaps. Besides, as the heteroatomic size increased from O to S and Se, the degree of intramolecular steric hindrance observed in the density functional theory calculations followed the trend of deviating from the coplanar backbone conformations. Consequently, the morphological and crystalline features of PFDPPF-Si were shown to be pronounced with a highly crystalline structure and dense π–π stacking, which led to the best hole mobility of 2.48 cm2 V−1 s−1. In addition, we demonstrated a complementary-like inverter based on PFDPPS-Si with a high gain of ∼54. The systematic characterization of heteroaromatics enables the design of conjugated copolymers, which provides a clear understanding of their structure–property behaviors on OFET characteristics.


 Please wait while we load your content...
Please wait while we load your content...