Copper-catalysed borylation of aryl chlorides†
Abstract
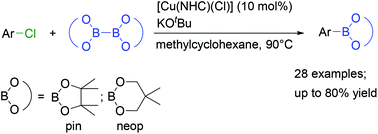
We report herein the first Cu-catalysed borylation of a wide range of aryl chlorides with different electronic and steric properties using a readily prepared NHC-stabilised Cu catalyst and KOtBu as the base with B2pin2 (pin = pinacolato) as the boron reagent. The aryl chlorides are converted into their corresponding arylboronic esters in good yields. The new procedure shows broad functional group tolerance, and B2neop2 (neop = neopentyl glycolato) can also be applied as the boron reagent.
- This article is part of the themed collections: Catalysis & biocatalysis in OBC and Trends in Organoboron Chemistry


 Please wait while we load your content...
Please wait while we load your content...