Colloidal Ni2−xCoxP nanocrystals for the hydrogen evolution reaction†
Abstract
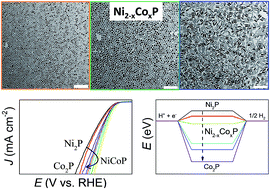
A cost-effective and scalable approach was developed to produce monodisperse Ni2−xCoxP nanocrystals (NCs) with composition tuned over the entire range (0 ≤ x ≤ 2). Ni2−xCoxP NCs were synthesized using low-cost, stable and low-toxicity triphenyl phosphite (TPP) as a phosphorus source, metal chlorides as metal precursors and hexadecylamine (HDA) as a ligand. The synthesis involved the nucleation of amorphous Ni–P and its posterior crystallization and simultaneous incorporation of Co. The composition, size and morphology of the Ni2−xCoxP NCs could be controlled simply by varying the ratio of Ni and Co precursors and the amounts of TPP and HDA. Ternary Ni2−xCoxP-based electrocatalysts exhibited enhanced electrocatalytic activity toward the hydrogen evolution reaction (HER) compared to binary phosphides. In particular, NiCoP electrocatalysts displayed the lowest overpotential of 97 mV at J = 10 mA cm−2 and an excellent long-term stability. DFT calculations of the Gibbs free energy for hydrogen adsorption at the surface of Ni2−xCoxP NCs showed NiCoP to have the most appropriate composition to optimize this parameter within the whole Ni2−xCoxP series. However, the hydrogen adsorption energy was demonstrated not to be the only parameter controlling the HER activity in Ni2−xCoxP.


 Please wait while we load your content...
Please wait while we load your content...