Formation of self-limited, stable and conductive interfaces between garnet electrolytes and lithium anodes for reversible lithium cycling in solid-state batteries†
Abstract
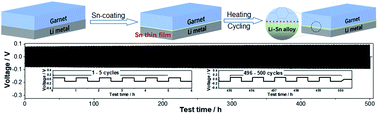
Solid-state batteries (SSBs) have already attracted significant attention due to their potential to offer high energy density and excellent safety as compared to the currently used lithium-ion batteries with liquid electrolytes. The use of a lithium anode in SSBs is extremely important to realize these advantages. Starting from the synthesis of a highly conductive cubic garnet solid electrolyte (Li6.375La3Zr1.375Nb0.625O12, LLZNO) using Nb as a structure stabilizer, in this study, we demonstrated the resolution of interfacial problems between the garnet electrolyte and lithium anode and the integration of the lithium anode into garnet-based SSBs by modifying the as-synthesized LLZNO with a Sn thin film. Due to the Sn modification, the interfacial resistances between the garnet electrolyte and the lithium anode decreased approximately 20 times to only 46.6 Ω cm2. The fast and reversible lithium plating/stripping under high current densities and the excellent battery performance of Li/Sn-LLZNO/LiFePO4 full cells were achieved. This improvement is ascribed to the formation of a Li–Sn alloy interlayer, which severs as a self-limited stable and conductive interface, bridging the garnet electrolyte and the lithium anode and enabling fast and stable lithium transport. As a proof-of-concept, this effective surface modification method will offer inspirations to researchers for overcoming the interfacial problems and promoting the development of high-performance SSBs.


 Please wait while we load your content...
Please wait while we load your content...