Heterometal functionalization yields improved energy density for charge carriers in nonaqueous redox flow batteries†
Abstract
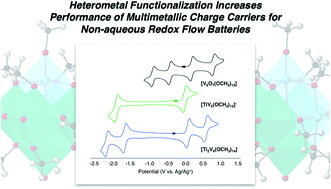
The development of facile, high-yielding synthetic routes to energy-dense charge carriers is critical for the success of redox flow battery technologies. Here, we present the results of synthetic modifications to our recently established series of polyoxovanadium clusters, [V6O7(OR)12] (R = CH3; C2H5), with respect to their performance as charge carriers for nonaqueous redox flow batteries. We demonstrate that incorporation of one ([TiV5(OCH3)13]−) or two ([Ti2V4(OCH3)14]) titanium ions within the Lindqvist core significantly increases the cell voltage of the system (from 1.60 V, to 2.30 and 2.74 V, respectively) while the solubility and redox stability observed for cluster complexes is retained. The improved physicochemical properties result in a 740% increase in energy density for [TiV5O6(OCH3)13]−, and a 210% increase for [Ti2V4O5(OCH3)14]. The kinetic implications of heterometal incorporation are assessed, demonstrating the importance of considering diffusion coefficients and heterogeneous electron transfer rate constants in mixed-metal charge carrier schematics. Ultimately, these results provide insight into structure–function relationships that will inform future synthetic design strategies of charge carriers for nonaqueous energy storage.


 Please wait while we load your content...
Please wait while we load your content...