In situ growth of single-layered α-Ni(OH)2 nanosheets on a carbon cloth for highly efficient electrocatalytic oxidation of urea†
Abstract
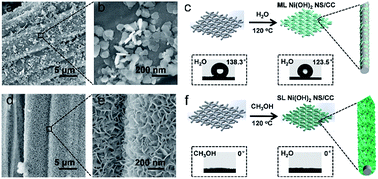
The electrochemical urea oxidation reaction (UOR) is useful for the simultaneous elimination of urea-rich wastewater and generation of hydrogen. In theory, nickel-based catalysts, as non-precious metal catalysts, show great advantages in this regard. However, poor conductivity and insufficient active sites on common nickel-based nanomaterials hinder their catalytic performance. Herein, we report methanol-directed one-step growth of single-layered (SL) α-Ni(OH)2 nanosheets on a conductive carbon cloth (CC) (SL α-Ni(OH)2 NS/CC) for highly efficient electrocatalytic urea oxidation. Without the use of any other pretreatment and modification methodology, it is found that huge SL α-Ni(OH)2 NS with a thickness of ∼0.8 nm densely and fully covers every CC fiber in the form of nanowalls. Taking advantage of the good conductivity of CC and the sufficiently exposed active surface of SL α-Ni(OH)2 NS, the resulting SL α-Ni(OH)2 NS/CC-integrated electrode displays superior UOR performance. A current density as high as 436.4 mA cm−2 at 0.5 V vs. Ag/AgCl is achieved, which represents the best UOR performance among those of the reported nickel-based electrocatalysts. This convenient approach opens a new way to obtain ultrafine heterogeneous catalysts and their integrated electrodes with the assistance of methanol solvent instead of water.


 Please wait while we load your content...
Please wait while we load your content...