Microenvironment alterations enhance photocurrents from photosystem I confined in supported lipid bilayers†
Abstract
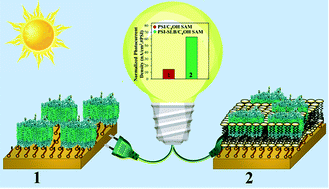
Transmembrane photosynthetic proteins, photosystem I (PSI), are nano-scale biological photodiodes that enable light-activated unidirectional electron flow. The robust photochemical properties of PSI make it a promising candidate for harnessing solar energy. However, the role of natural membrane confinements of PSI in orchestrating this photoactivated charge separation with near unity quantum efficiency, which is central to the rational design of PSI-based energy conversion systems, is still ill-understood. Motivated by this lack of fundamental understanding, herein we investigate the photoactivity of biomimetic constructs of cyanobacterial PSI encapsulated within solid-supported lipid bilayers (SLB) assembled on electrodes. PSI confined in SLBs is assembled from PSI-proteoliposomes that are synthesized from our recently developed facile routes for engineering negatively charged phospholipid (DPhPG) bilayer membranes. Specifically, detailed chronoamperometry measurements have been used to investigate photocurrent variations arising from the SLBs supported on self-assembled monolayer (SAM) substrates. These measurements, in conjunction with cryo-transmission electron microscopy, atomic force microscopy imaging and force spectroscopy, allow for direct visualization and detection of the SLBs of PSI-proteoliposomes on the substrates. Our results indicate the critical role of microenvironment alterations, heretofore not considered, in achieving ∼4–5 fold enhancements in photocurrents generated from PSI complexes under SLB confinements as compared to those from a dense monolayer of equivalent concentrations of PSI on SAM substrates.


 Please wait while we load your content...
Please wait while we load your content...