Calixarene alpha-ketoacetylenes: versatile platforms for reaction with hydrazine nucleophile†
Abstract
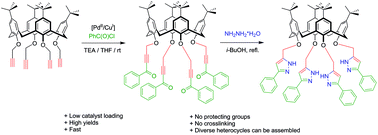
Late stage diversification of calix[4]arenes and thiacalix[4]arenes with heterocycles remains a significant synthetic challenge and hampers further exploitation of the scaffolds. Here we describe the development of a short and facile synthetic route to conformationally diverse novel calix[4]arene and thiacalix[4]arene ynones using a palladium cross coupling approach (5% Pd(II) + 10% Cu(I)) with benzoyl chloride. Their successful conversion to heterocycles to afford pyrazoles was demonstrated through treatment with hydrazine. Functionalisation is calixarene conformation and linker independent enabling access to a library of structures.


 Please wait while we load your content...
Please wait while we load your content...