Porphyrin-based porous polyimide polymer/Pd nanoparticle composites as efficient catalysts for Suzuki–Miyaura coupling reactions†
Abstract
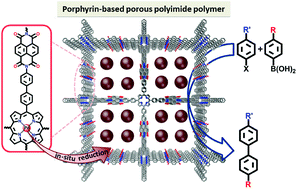
Two porphyrin-based porous polyimide polymers, PPPP-1 and PPPP-2, were synthesized via 4 + 2 polyimide reactions of extended porphyrin tetraamines (TBPP) and pyromellitic dianhydride (PMDA), or naphthalene dianhydride (NTDA). The polyimide structures were unambiguously confirmed by FT-IR and 13C CP-MAS solid state NMR. Powder X-ray diffraction and computer modeling analysis confirmed the crystallinity and the layered structures with tetragonal channels of PPPP-1 and PPPP-2. The Brunauer–Emmett–Teller (BET) surface area together with pore size distribution suggested permanent porosity, whose surface areas were calculated as 295.07 m2 g−1 and 301.48 m2 g−1 for PPPP-1 and PPPP-2, respectively. Furthermore, the morphology of PPPPs was confirmed by exhaustive scanning electron microscopy (SEM) and transmission electron microscopy (TEM) imaging. Highly dispersed Pd nanoparticles with a diameter of 3 ± 2 nm were successfully immobilized into PPPPs through the coordination of Pd(II) ions with porphyrin units on polymers and in situ reduction afterwards. Pd@PPPP-1 and Pd@PPPP-2 show significantly excellent catalytic activity in Suzuki–Miyaura coupling reactions. The performance of Pd@PPPPs is apparently superior to that of the homogeneous palladium catalyst. In addition, Pd@PPPPs have excellent stability and recyclability, and they can be reused without loss of activity in five successive reaction cycles. These results indicate that the polyimide porphyrin porous organic polymers are promising candidates for efficient heterogeneous catalysis.


 Please wait while we load your content...
Please wait while we load your content...