Thermally induced cationic polymerization of isobutyl vinyl ether in toluene in the presence of solvate ionic liquid†
Abstract
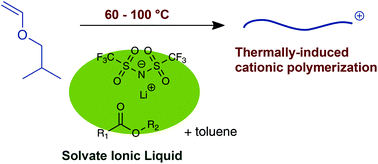
Radical polymerization of isobutyl vinyl ether (IBVE) was attempted with the aid of the interaction between the corresponding propagating radical and lithium cation (Li+). LiN(SO2CF3)2 (LiNTf2) and ester compounds, such as methyl methacrylate (MMA) and vinyl acetate (VAc), were added as a Li+ source and dissolving agent for LiNTf2, respectively. Homopolymers of cationically polymerizable IBVE were obtained despite the presence of radically polymerizable monomers such as MMA and VAc. Contrary to our expectation, the polymerization proceeded via not a radical mechanism but a cationic mechanism. However, this cationic polymerization was found to be unusual. In particular, the polymer yield increased with the polymerization temperature; successful polymerization was observed at 100 °C, whereas no polymerization occurred at lower temperatures such as at 0 °C. The behavior of the present system was therefore defined as “thermally induced cationic polymerization”. The mechanism of thermally induced cationic polymerization is still not clear, but it is assumed that the propagating cation is markedly stabilized through its interaction with the solvate ionic liquid formed between LiNTf2 and the Lewis base.


 Please wait while we load your content...
Please wait while we load your content...