Mild and efficient synthesis of indoles and isoquinolones via a nickel-catalyzed Larock-type heteroannulation reaction†
Abstract
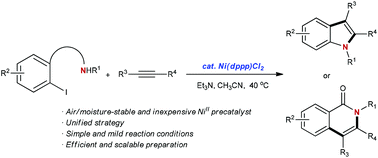
A simple and efficient approach for the preparation of substituted indoles and isoquinolones via a nickel-catalyzed Larock-type heteroannulation reaction is reported. This transformation employed air-stable and inexpensive Ni(dppp)Cl2 as a precatalyst and Et3N as a mild base. Moreover, the reaction occurs efficiently under mild conditions, and a wide range of substituted indoles and isoquinolones bearing various functional groups are obtained in moderate to excellent yields.
- This article is part of the themed collection: Synthetic methodology in OBC


 Please wait while we load your content...
Please wait while we load your content...