A catalyst-controlled switchable reaction of β-keto acids to silyl glyoxylates†
Abstract
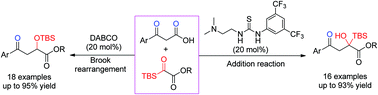
A catalyst-controlled switchable reaction of β-keto acids to silyl glyoxylates was developed under mild conditions, providing two distinct products in good yields. Compared to decarboxylative addition-Brook rearrangement, the decarboxylative addition products could be controlled by using a simple bifunctional organocatalyst thiourea derivative instead of DABCO. This new reaction model of silyl glyoxylates offered a facile and alternative route to organosilicon compounds.
- This article is part of the themed collection: Synthetic methodology in OBC


 Please wait while we load your content...
Please wait while we load your content...