Design and synthesis of (aza)indolyl maleimide-based covalent inhibitors of glycogen synthase kinase 3β†
Abstract
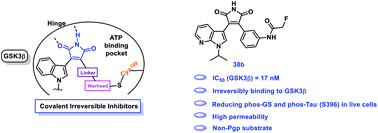
As an important kinase in multiple signal transduction pathways, GSK-3β has been an attractive target for chemical probe discovery and drug development. Compared to numerous reversible inhibitors that have been developed, covalent inhibitors of GSK-3β are noticeably lacking. Here, we report the discovery of a series of covalent GSK-3β inhibitors by optimizing both non-covalent interactions and reactive groups. Among these covalent inhibitors, compound 38b with a mild α-fluoromethyl amide reactive group emerges as a selective and covalent inhibitor against GSK-3β, effectively inhibits the phosphorylation of glycogen synthase and tau protein, and increases β-catenin's levels in living cells. In addition, compound 38b is highly permeable and not a substrate of P-glycoprotein.


 Please wait while we load your content...
Please wait while we load your content...