New amyloid beta-disaggregating agents: synthesis, pharmacological evaluation, crystal structure and molecular docking of N-(4-((7-chloroquinolin-4-yl)oxy)-3-ethoxybenzyl)amines†
Abstract
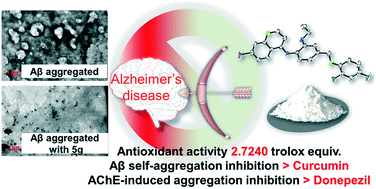
In the journey towards the development of potent multi-targeted ligands for the treatment of Alzheimer's disease, a series of Aβ aggregation inhibitors having quinoline scaffold were designed utilizing computational biology tools, synthesized and characterized by various spectral techniques including single-crystal X-ray crystallography. Organic syntheses relying upon convergent synthetic routes were employed. Investigations via ThT fluorescence assay, electron microscopy and transmission electron microscopy revealed the synthesized derivatives to exhibit Aβ self-aggregation inhibition. Molecules 5g and 5a showed the highest inhibitory potential, 53.73% and 53.63% at 50 μM respectively; higher than the standard Aβ disaggregating agent, curcumin. Molecules 5g and 5a disaggregated AChE-induced (58.26%, 47.36%) Aβ aggregation more than two fold more than the standard drug-donepezil (23.66%) and inhibited Cu2+-induced Aβ aggregation. A docking study significantly showed their interaction with key residues of Aβ and the results were in accordance with the study. Besides, these compounds also exhibited potential antioxidant activity (5a, 2.7240 Trolox equivalent by ORAC assay) and metal chelating property. Furthermore, the stoichiometric ratio of Cu (II)–5a and Cu(II)–5g complexes were found by Job's method (0.5 : 1 for 5a and 0.8 : 1 for 5g). In silico ADMET profiling showed these derivatives to have drug like properties with very low toxicity effects in the pharmacokinetic study. Overall, these results displayed a multi-activity profile with promising Aβ aggregation inhibition and antioxidation and metal chelation activity that could be helpful for developing new multifunctional agents against Alzheimer's disease.


 Please wait while we load your content...
Please wait while we load your content...