Extended visible to near-infrared harvesting of earth-abundant FeS2–TiO2 heterostructures for highly active photocatalytic hydrogen evolution†
Abstract
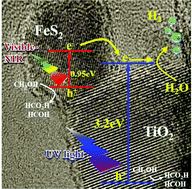
Photocatalytic water splitting is a key technology for long-term hydrogen evolution with low environmental impact. In this work, an environment-friendly photocatalyst consisting of FeS2–TiO2 heterostructured nanocrystals has been successfully prepared by wet chemical synthesis for enhancing the photocatalytic hydrogen production rate over a wide range of absorption wavelengths. Its absorption range has been extended from the ultraviolet–visible to near-infrared (NIR) spectrum. The results of ultrafast optical spectroscopy suggest that charge transfer occurs between the FeS2 and TiO2 interface under NIR pulsed laser irradiation (800 nm). The highest photocatalytic hydrogen production rate of the FeS2–TiO2 heterostructure has been obtained in comparison with FeS2 nanocrystals (NCs) and TiO2 nanoparticles (NPs) under irradiation by a mercury arc lamp or a xenon lamp. In the FeS2–TiO2 heterostructure, the FeS2 NCs as a light harvester can efficiently absorb visible and NIR light to generate much more photoelectrons than TiO2 NPs only for hydrogen production. For long-term photocatalytic hydrogen production, the FeS2–TiO2 heterostructures also reveal an outstanding durability over 40 h. Overall, the FeS2–TiO2 heterostructure shows potential to be a TiO2-based photocatalyst for clean hydrogen production in practical applications.


 Please wait while we load your content...
Please wait while we load your content...