Kinetic and equilibrium adsorption of lead from water using magnetic metformin-substituted SBA-15
Abstract
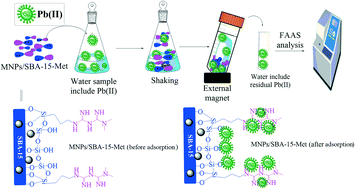
The present investigation demonstrates the successful and easy approach for the synthesis of magnetic amine-substituted SBA-15 nanocomposite for effective removal of lead (Pb2+) ions via adsorption from aqueous environment. The nano-adsorbents were synthesized by effective doping of magnetic nanoparticles over synthesized SBA-15 metformin (Met) to obtain MNPs/SBA-15-Met nanocomposites. The synthesized nanocomposites were characterized by Fourier transform infrared spectroscopy (FT-IR), EDX, X-ray diffraction (XRD) and thermogravimetric analysis (TGA). Due to the presence of nitrogen atom containing backbone, the MNPs-SBA-15-Met nanocomposite has demonstrated high adsorption capacity (333 mg g−1) toward Pb(II) ions as compared to the pure SBA-15. Experimental data was evaluated with isotherm models and non-linear type I model was well fitted on data. The effective parameters for adsorption process such as effect of pH, salt, adsorbent dosage, contact time, initial concentration and temperature were studied and optimized. The studies indicated that at pH range of 6, the percentage removal of Pb(II) was found to be 96% at room temperature. The FTIR spectroscopy and EDX results of the nanocomposites after the Pb(II) adsorption (MNPs/SBA-15-Met-Pb2+) specified that adsorption occurred probably through electrostatic interaction between Pb(II) ions and N groups (amine & amide).


 Please wait while we load your content...
Please wait while we load your content...