Emerging investigator series: sunlight photolysis of 2,4-D herbicides in systems simulating leaf surfaces†
Abstract
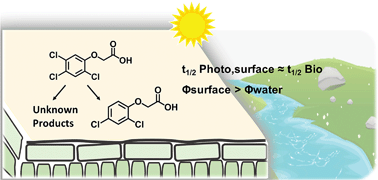
Pesticides are commonly applied on foliage, forming dry deposits on the leaf cuticular wax. However, their photochemical transformation in this lipophilic environment is much less understood compared with that in surface water. In this work, sunlight photolysis of six chlorinated phenoxyacetic acid herbicides (i.e., 2,4-D and structural analogues) was evaluated in four organic solvents, on quartz, and on paraffin wax. In solvents of low polarity (i.e., n-heptane and 2-propanol), direct photolysis of 2,4-D herbicides was enhanced due to the relatively high quantum yields in these solvents. Photolysis on paraffin wax was slower than photolysis on quartz by a factor of 3–9, but was comparable with that in solvents of low polarity. With environmentally relevant irradiation and surface loading, the half-lives of 2,4-D herbicides on paraffin wax were 27–159 h, which are within the same range reported for biodegradation, the dominant dissipation pathway in the current 2,4-D fate model. Product analyses showed that photoreductive dechlorination is the dominant pathway in organic solvents, accounting for 68–100% of parent compound decay. On quartz and paraffin wax surfaces, however, photoreductive dechlorination products accounted for <60% of parent compound decay. Combining kinetic modeling and product analyses, it was shown that neither could the two additional putative pathways (photosubstitution of chlorine by hydroxyl group and cleavage of the ether bond) fully account for the total phototransformation on surfaces. These results suggest that rapid photolysis on surfaces can be attributed to unique pathways that are absent in the organic solvent phase.
- This article is part of the themed collection: Emerging Investigator Series


 Please wait while we load your content...
Please wait while we load your content...