A 2,2′-bipyridine-containing covalent organic framework bearing rhenium(i) tricarbonyl moieties for CO2 reduction†
Abstract
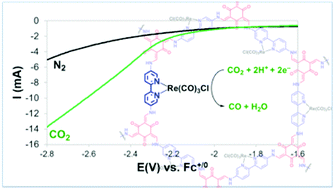
The reduction of CO2 into higher energy products such as carbon-based fuels and feedstocks is an attractive strategy for mitigating the continuous rise in CO2 emissions associated with the growing global energy demand. Rhenium tricarbonyl complexes bearing 2,2′-bipyridine (2,2′-bpy) ligands are well-established molecular electrocatalysts for the selective reduction of CO2 to CO. Construction of efficient devices for this electrochemical process requires the immobilization of electrocatalysts to electrode surfaces. To integrate Re(2,2′-bpy)(CO)3 fragments into a covalent organic framework (COF), Re(5,5′-diamine-2,2′-bpy)(CO)3Cl (1) was synthesized and electrochemically investigated. Complex 1 is an active and selective electrocatalyst for the reduction of CO2 to CO with excellent faradaic efficiency (99%). The presence of the amine substituents leads to a destabilization of the π* orbital of the 5,5′-diamine-2,2′-bpy ligand with respect to the metal center. Therefore, 1 requires more negative potentials (−2.47 V vs. Fc+/0) to reach the doubly reduced catalytically active species. DFT studies were conducted to understand the electronic structure of 1, and support the destabilizing effect of the amine substituents. The Re-2,2′-bpy fragments were successfully integrated into a COF containing 2,2′-bpy moieties (COF-2,2′-bpy) via a post-metallation synthetic route to generate COF-2,2′-bpy-Re. A composite of COF-2,2′-bpy-Re, carbon black, and polyvinylidene fluoride (PVDF) was readily immobilized onto glassy carbon electrodes and electrocatalytic CO2 reduction to CO was observed at −2.8 V vs. Fc0/+, with a faradaic efficiency of 81% for CO production.


 Please wait while we load your content...
Please wait while we load your content...