Single-atom iron catalyst with single-vacancy graphene-based substrate as a novel catalyst for NO oxidation: a theoretical study†
Abstract
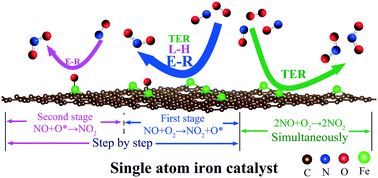
Nitric oxide (NO) emitted from coal-fired power plants has raised global concerns. The catalytic oxidation of NO is a promising technology for NO removal. Due to its high selectivity and high catalytic activity, a single-atom iron catalyst supported on a single-vacancy (SV) graphene-based substrate (Fe/SV) may be a promising catalyst for NO oxidation. The adsorption characteristics and reaction mechanisms of NO and O2 on the surface of Fe/SV were systematically studied through density functional theory calculation. The detailed reaction paths under the Langmuir–Hinshelwood, Eley–Rideal and termolecular Eley–Rideal reaction mechanisms were investigated, and thermodynamic and kinetic analyses were performed to study the effect of reaction temperature on the reaction rate and equilibrium. It is found that there are six different reaction paths which can be divided into two reaction patterns, step-by-step and simultaneous. The minimum activation energy of NO oxidation is only 0.04 eV and Eley–Rideal seems to be the dominant reaction mechanism of NO oxidation in flue gas. The reaction temperature has an obvious promoting effect on the reaction rate and an inhibitory effect on the reaction equilibrium. We hope that this research can lay a foundation for further theoretical research on NO oxidation and provide a guideline for experimental investigation on NO oxidation.


 Please wait while we load your content...
Please wait while we load your content...