Diffusion mechanism of Na ion–polaron complex in potential cathode materials NaVOPO4 and VOPO4 for rechargeable sodium-ion batteries†
Abstract
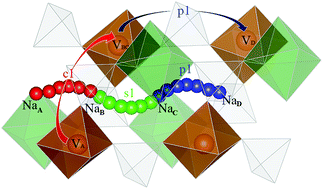
Using the density functional method, we investigated the crystal and electronic structures and the electrochemical properties of NaxVOPO4 (x = 0, 1) and explored the diffusion mechanism of Na ions in these materials. The van der Waals interaction was also taken into account to include the non-local electron correlation in the calculation of structural parameters and voltage. The diffusion of Na ions is treated as a process of the Na vacancy–positive small polaron complex in NaVOPO4 and the Na ion–negative small polaron complex in VOPO4, respectively. During the charging (discharging) process, the removal (insertion) of a Na ion would result in the formation of a positive (negative) small polaron at one of the two first nearest vanadium sites to the Na vacancy. Three elementary diffusion processes, including the single, crossing and parallel diffusion processes, are explored. It is found that the [010] direction is preferable for Na ion diffusion in both the charging and discharging processes. The influence of small polaron migration on Na ion diffusion in the charging state is negligible, whereas such effect is considerably strong in the discharging process. Moreover, while three elementary diffusion processes in NaVOPO4 require the same energy, the parallel diffusion process in VOPO4 is not preferred. The diffusion of Na vacancy accompanied by a positive polaron in the full charging process requires an activation energy of 395 meV, while the diffusion of Na ion accompanied by a negative polaron in the discharging state, VOPO4, has a higher activation energy of 627 meV. With a voltage and activation barrier similar to that of the olivine phosphate LiFePO4, these sodium-based materials are expected to be promising cathode materials for sodium ion batteries.


 Please wait while we load your content...
Please wait while we load your content...