SO2 absorption in EmimCl–TEG deep eutectic solvents†
Abstract
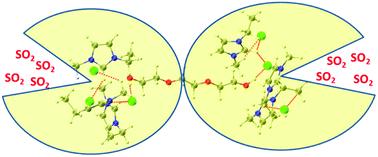
Deep eutectic solvents (DESs) based on 1-ethyl-3-methylimidazolium chloride (EmimCl) and triethylene glycol (TEG) with different molar ratios (from 6 : 1 to 1 : 1) were prepared. FTIR and theoretical calculation indicated that the C2–H on the imidazolium ring form hydrogen bonds with the hydroxyl group rather than the ether O atom of the TEG. The EmimCl–TEG DESs can efficiently capture SO2; in particular, EmimCl–TEG (6 : 1) can capture 0.54 g SO2 per gram of solvent at 0.10 atm and 20 °C, the highest absorption amount for DESs under the same conditions. Theoretical calculation showed that the high SO2 absorption capacity was mainly due to the strong charge-transfer interaction between SO2 and the anion Cl−. Moreover, SO2 desorption in the DESs can be controlled by tuning the interaction between EmimCl and TEG, and the DESs can be cycled many times.


 Please wait while we load your content...
Please wait while we load your content...