Inhibition of carbonic anhydrases by a substrate analog: benzyl carbamate directly coordinates the catalytic zinc ion mimicking bicarbonate binding†
Abstract
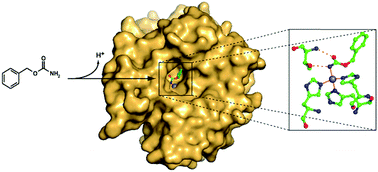
N-Unsubstituted carbamates have scarcely been investigated so far as carbonic anhydrase inhibitors (CAIs). By means of kinetic and structural studies, in this paper we demonstrate that such molecules can effectively inhibit hCAs and can be used as lead compounds for the development of CAIs possessing a binding mode similar to one of the CA substrates, bicarbonate.


 Please wait while we load your content...
Please wait while we load your content...