Reduction-responsive amphiphilic polymeric prodrugs of camptothecin–polyphosphoester for cancer chemotherapy†
Abstract
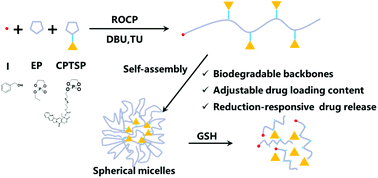
As one of the promising prodrug strategies, amphiphilic polymeric prodrugs can be constructed by conjugating some small molecular hydrophobic anticancer drugs onto a hydrophilic biodegradable and biocompatible polymer, which further self-assembles into nanoparticles with controllable drug loading. Here, a biocompatible and biodegradable polyphosphoester and a broad-spectrum anticancer drug camptothecin (CPT) were selected as model compounds. We first designed and synthesized a novel cyclic phosphoester monomer (CPTSP) containing a disulfide bond linker and CPT as a side group. Then a reduction-responsive amphiphilic polymeric prodrug (PCPTSP-co-PEEP) was prepared through the ring-opening copolymerization of CPTSP and a common cyclic phosphoester monomer containing ethyl (EEP) as the side group, which was initiated by benzyl alcohol with 1,8-diazabicycloundec-7-ene (DBU) and thiourea (TU) as catalysts. The resulting PCPTSP-co-PEEPs could self-assemble into micelles in water due to their hydrophilic polyphosphoester main chain and some hydrophobic CPT side groups. The average size and the drug loading content of these micelles can be controlled by adjusting the feed molar ratio of EEP and CPTSP. The average hydrodynamic diameter increased from 190 nm to 250 nm when the molar ratio of EEP/CPTSP was adjusted from 9/1 to 49/1. These micelles possessed appropriate stability under physiological conditions and degraded in the reduction medium according to their change in size monitored by DLS measurement. Their reduction-responsive properties were also confirmed by the in vitro drug release behavior. Moreover, these micelles could enter into HT29 tumor cells via endocytosis and released CPT efficiently to inhibit their proliferation. The in vivo tumor inhibition rate of these polymeric prodrugs was 73.4%, which was about two fold that for free CPT (36.5%). This approach of polymeric prodrugs based on polyphosphoesters can be extended to other anticancer drugs to construct smart drug delivery systems for cancer treatment.


 Please wait while we load your content...
Please wait while we load your content...