Osteogenic nanostructured titanium surfaces with antibacterial properties under conditions that mimic the dynamic situation in the oral cavity†
Abstract
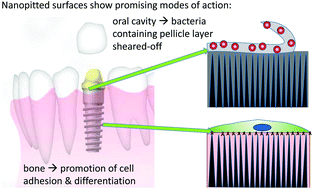
The study aim was to assess the impact of different surface nanofeatures on otherwise smooth titanium surfaces on bacterial adhesion as well as on their osteogenic potential. Bacterial adhesion was assessed in the presence of saliva under static and dynamic conditions to approximate both sub- and supragingival conditions in the oral cavity as the gingival seal will be affected by implantation. The ultimate goal was to develop a surface that will reduce biofilm formation but still support osseointegration in vivo. To this end nanotubular or nanopitted surfaces were created on electropolished titanium via electrochemical anodization procedures. Sandblasted/acid etched surfaces (SBAE) were used as a microrough reference. Bacterial adhesion was studied using saliva-precoated samples with S. sanguinis as a typical early colonizer of the oral cavity; osteogenic differentiation was assessed with human bone marrow stromal cells. While bacterial adhesion was reduced on all microsmooth surfaces to an average of 17% surface coverage compared to 61% on SBAE under static conditions, under dynamic conditions the nanopitted surface had a significant impact on bacterial adhesion. Here fluid flow removed all bacteria. By comparison, the reduction on the nanotubular surface was only similar to that of the SBAE reference. We hypothesise the underlying cause to be an effect of the surface morphology on the structure and composition of the saliva precoating that reduces its stability, giving rise to a self-cleaning effect. In addition, no negative influence on the osteogenic potential of the nanopitted surface could be determined by alkaline phosphatase activity, mineralization behaviour or gene expression; it remained on a par with the tissue culture plastic control. Thus, nanopitting seems to be a promising surface treatment candidate for dental implants to reduce infection related complications without compromising the implant integration.


 Please wait while we load your content...
Please wait while we load your content...