A new functional membrane protein microarray based on tethered phospholipid bilayers†
Abstract
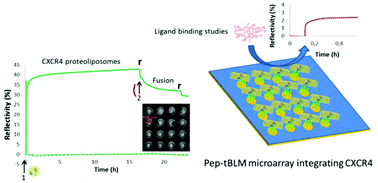
A new prototype of a membrane protein biochip is presented in this article. This biochip was created by the combination of novel technologies of peptide-tethered bilayer lipid membrane (pep-tBLM) formation and solid support micropatterning. Pep-tBLMs integrating a membrane protein were obtained in the form of microarrays on a gold chip. The formation of the microspots was visualized in real-time by surface plasmon resonance imaging (SPRi) and the functionality of a GPCR (CXCR4), reinserted locally into microwells, was assessed by ligand binding studies. In brief, to achieve micropatterning, P19-4H, a 4 histidine-possessing peptide spacer, was spotted inside microwells obtained on polystyrene-coated gold, and Ni-chelating proteoliposomes were injected into the reaction chamber. Proteoliposome binding to the peptide was based on metal–chelate interaction. The peptide-tethered lipid bilayer was finally obtained by addition of a fusogenic peptide (AH peptide) to promote proteoliposome fusion. The CXCR4 pep-tBLM microarray was characterized by surface plasmon resonance imaging (SPRi) throughout the building-up process. This new generation of membrane protein biochip represents a promising method of developing a screening tool for drug discovery.

 Please wait while we load your content...
Please wait while we load your content...