Clear distinction between CAC and CMC revealed by high-resolution NMR diffusometry for a series of bis-imidazolium gemini surfactants in aqueous solutions†
Abstract
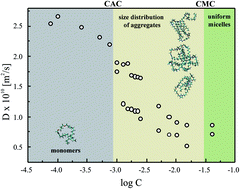
The aggregation behavior in the transition region was studied for a series of dicationic surfactants 3,3′-[α,ω-(dioxaalkane)]bis(1-dodecylimidazolium)dichlorides with varied spacer length from two to twelve carbon atoms. We employed Nuclear Magnetic Resonance diffusometry and Bayesian DOSY analysis to obtain the aggregate size distribution in the transition region. The critical concentrations CC were independently obtained from surface tension, electric conductivity, UV-Vis and NMR methods. The micelle aggregation numbers were estimated from the self-diffusion coefficients and were independently confirmed using steady-state fluorescence quenching. The morphology of the aggregates was characterized by small-angle scattering of synchrotron radiation and molecular dynamics simulations. The obtained CC values are identified as critical aggregation concentrations CAC. A broad transition region was observed, and stable micelles were obtained at much higher concentrations than CAC. The accurate CMC values could not be identified for the systems in the study. We indicated that the distribution of aggregate size becomes small and the system becomes homogeneous at much larger concentrations than CAC (typically 15–20 mM). The existence of a slow exchange between two environments, an aggregate and aqueous environment, was confirmed by 1H NMR and 2D HSQC NMR spectroscopy.


 Please wait while we load your content...
Please wait while we load your content...