Simple low cost porphyrinic photosensitizers for large scale chemoselective oxidation of sulfides to sulfoxides under green conditions: targeted protonation of porphyrins†
Abstract
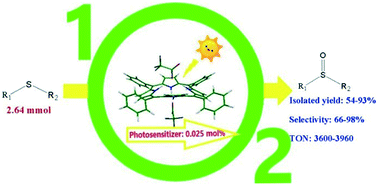
Aerobic photooxidation of sulfides with the diacids of meso-tetra(phenyl)porphyrin, H2TPP (acid = CF3COOH, Cl2CHCOOH, HClO4 and H2SO4) under sunlight in acetonitrile gave the corresponding sulfoxides as nearly the exclusive product. The photocatalytic activity of the dications decreased as H4TPP(ClO4)2 ≫ H4TPP(Cl2CHCOO)2 ≫ H4TPP(CF3COO)2 > H4TPP(HSO4)2. Also, H4TPP(ClO4)2 and H4TPP(Cl2CHCOO)2 showed a remarkably increased oxidative stability in comparison with the free base porphyrin. The singlet oxygen quantum yield (φΔ) of the porphyrin diacids was found to be dependent on the acid used for the protonation of H2TPP. The solvent effect on the efficiency of the oxidation reaction was investigated using acetonitrile, 1,2-dichloroethane (DCE) and toluene. The higher conversions achieved in acetonitrile and DCE are in accord with the higher singlet oxygen lifetime in these solvents. Also, the lower fluorescence quantum yield of porphyrin dications in acetonitrile relative to that in DCE was used to explain the higher efficiency of the former. The porphyrin diacids can also accelerate the oxidation reaction through an acid catalysis mechanism. Also, cooxidation of methyl phenyl sulfide and diphenyl sulfide was conducted to provide insights into the nature of the active intermediates involved in the oxidation of sulfides to sulfoxides and partial overoxidation of the sulfoxides to sulfones. The results showed much higher reactivity of the persulfoxide intermediate compared to that of singlet oxygen. The catalytic system was successfully employed to the large scale preparation of sulfoxides from the corresponding sulfides under mild conditions.


 Please wait while we load your content...
Please wait while we load your content...