Phosphorus anionic redox activity revealed by operando P K-edge X-ray absorption spectroscopy on diphosphonate-based conversion materials in Li-ion batteries†
Abstract
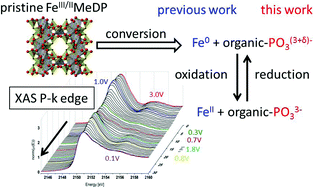
When cycling diphosphonate-based organic–inorganic hybrid materials as negative battery electrodes, specific charges exceeding the maximum for a metal redox reaction are recorded. Classical explanations are electrolyte reduction and solid electrolyte interphase (SEI) oxidation. Using operando X-ray absorption spectroscopy (XAS) at the P K-edge experiment, we demonstrate an additional contribution of reversible ligand co-cycling based on the P-atoms of the diphosphonate ligands, upon delithiation occurring during the first potential plateau, which matches perfectly to previous investigations using Fe K-edge XAS. We thus investigate experimentally an anionic redox process in situ. Thereby we could demonstrate a possible methodology to measure P K-edge XANES on Li-ion battery active materials under operando conditions, which could be applied to verify the predicted P co-cycling of other conversion-type negative electrode materials.


 Please wait while we load your content...
Please wait while we load your content...