Is nano ZrO2 a better photocatalyst than nano TiO2 for degradation of plastics?
Abstract
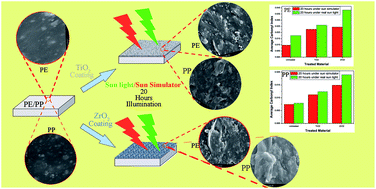
The environmental accumulation of plastic is a huge problem due to its low degradability. There are solutions to this problem such as reusing, recycling and the use of biodegradable plastics. However, a complete solution to this problem has not yet been achieved. In this study, photocatalytic degradation of polyethylene and polyropylene was investigated. The effect of ZrO2 nanoparticles in comparison with TiO2 nanoparticles for photocatalytic degradation was studied. TiO2 nanoparticles were synthesized by a sol gel method and ZrO2 nanoparticles were synthesized by a sonochemical method. The TiO2 and ZrO2 particles were characterized using FTIR, XRD, UV visible spectroscopy, EDX, SEM, and TEM. Both types of particles were approximately 50 nm in size. TiO2 nanoparticles were tetragonal and in the anatase phase. ZrO2 particles were tetragonal and nano porous. The application of these particles to polyethylene and polypropylene was performed using nanoparticle suspensions in a THF medium. The degradation of the plastics was studied by investigating chemical changes using FTIR and morphological changes using SEM. Optimization of the concentration and exposure time was performed under laboratory conditions using a sun simulator. Polyethylene and polypropylene were treated under the sun simulator as well as under the real sun light conditions. In both treatment conditions, it was found that there is a significant difference in the degradation of plastics and ZrO2 nanoparticle suspension treated polyethylene and polypropylene showed higher degradation than the TiO2 nanoparticle suspension treated samples at 95% confidence levels.


 Please wait while we load your content...
Please wait while we load your content...