Breaking the disulfide chemical bond using high energy photons: the dimethyl disulfide and methyl propyl disulfide molecules
Abstract
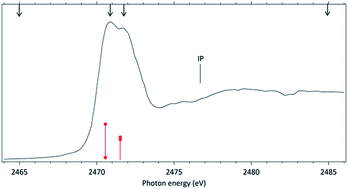
In order to study the stability of the disulfide chemical bond in molecules subjected to a flux of high energy photons, the ionic fragmentation of two molecules containing a disulfide chemical bond, dimethyl disulfide (DMDS) and methyl propyl disulfide (MPDS), has been studied following excitation around the S 1s edge (∼2470 eV). Synchrotron radiation and electron-ion coincidence techniques were used. The core excited states of DMDS and MPDS have also been theoretically investigated at the multiconfigurational self-consistent field (MCSCF) level and multireference perturbation theory. Deep core excitation (S 1s) induces multiple ionization and intense fragmentation of the molecules, as clearly demonstrated by the observation of cations such as S+, S2+ and S3+. Splitting of the S+ peak is observed in the mass spectra of the two molecules when they are excited to a resonant state with antibonding (S–S) character. Although fragments associated with the breakage of the disulfide bond (S+ and CHnS+) play a dominant role in the dissociation of the molecules, a fragment containing the disulfide chemical bond, S2+, survives, nevertheless, at all photon energies.


 Please wait while we load your content...
Please wait while we load your content...